Events
Plastic Surgery The Meeting – 2025 – BOOTH 1912
October 9-12, 2025

IN THE NEWS
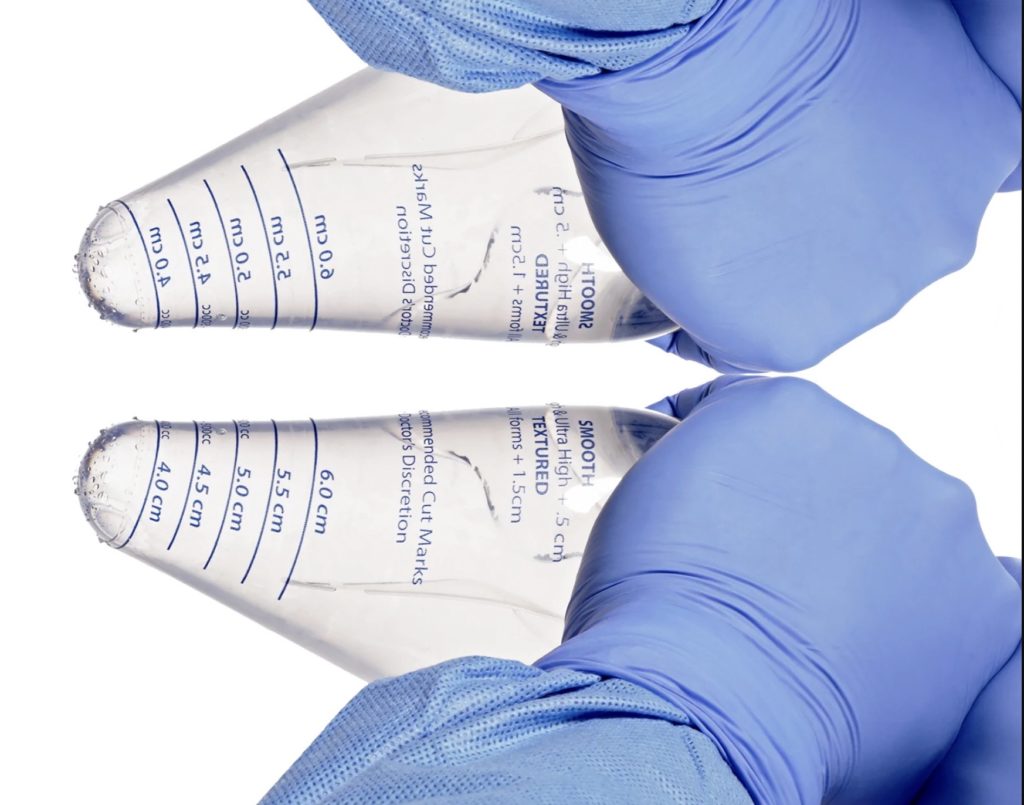
Safety of a Protective Funnel in Primary Breast Augmentation: A Retrospective Analysis of 380 Multicenter Cases in the United States
April 17, 2021
Protective funnel devices are commonly used to deliver implants in primary breast augmentation (BA) yet there is a paucity of evidence-based data describing their safety in the literature.
Proximate Concepts Receives ISO 13485:2016 Certification, Expands Education and Scholarly Publications With iNPLANT Funnel
March 17, 2021
iNPLANT LLC, a medical device company and manufacturer of the iNPLANT Funnel, announced today that its parent company, Proximate Concepts LLC, has been awarded ISO13485:2016 certification for Medical Device and Quality Management Systems from SGS, the world’s leading inspection, verification, testing and certification company. – Continue Reading on PRNEWSWIRE.com
March 17, 2021
iNPLANT LLC, a medical device company and manufacturer of the iNPLANT Funnel, announced today that its parent company, Proximate Concepts LLC, has been awarded ISO13485:2016 certification for Medical Device and Quality Management Systems from SGS, the world’s leading inspection, verification, testing and certification company. – Continue Reading on PRNEWSWIRE.com
Double Loading of Breast Implants in Aesthetic and Reconstructive Plastic Surgery With the iNPLANT Funnel
March 12, 2021
According to The Aesthetic Society’s annual statistics report, breast augmentation (BA) procedures continually rank among the most performed procedures, with a total of 280,692 performed in 2019 by 87.3% of plastic surgeons certified by the American Board of Plastic Surgery.
– Continue Reading on OUP.com
PRIVATE EVENTS
Take a Personal or Virtual Tour of our Medical Instrument Museum
Schedule a webinar +1 (201) 913-8833
Take a virtual tour of our medical antique collection at inplant toolsofyesterday.org






What Surgeons Are Saying
recognition